Introduction to integrated methods in the vegetable garden
chapter crop sol
Influence of pH on the fertility potential of cultivated soils
Click on ♦ to go to a page in the chapter
Analysis of the physico-chemical properties of cultivated soils ♦
. Texture and structure of cultivated soils ♦
. Clay-humus complexes and cation exchange capacity ♦
. Other interesting data that may be included in a laboratory analysis ♦
⇒ Influence of pH on the fertility potential of cultivated soils
Humus; formation and evolution ♦
Soil fertility: is the apocalypse coming tomorrow? ♦
The microbial world and soil fertility ♦
Rhizosphere, mychorizae and suppressive soils ♦
Correction of soils that are very clayey, too calcareous or too sandy ♦
Stimation of humus losses in cultivated soil ♦
Compost production for a vegetable garden ♦
Composting with thermophilic phase ♦
Weed management in the vegetable garden ♦
To plow or not to plow? ♦
The rotary tiller, the spade fork, and the broadfork ♦
Please share this website with your friends and family and anyone else who might be interested in integrated farming methods.
The pH (potential hydrogen) describes the acidity or alkalinity of the soil solution. Soils are considered to be:
- Moderately acidic when the pH is below 6, and highly acidic when the pH is below 5.
- Moderately basic (alkaline) when the pH is above 8 and very basic when the pH is above 9.
- Neutral when pH = 7
Soil that is low in limestone tends to become acidic (pH < 7). Soil that is rich in limestone tends to be alkaline (pH > 7).
The pH value of the soil has a significant effect on biomass. Significant changes in pH can result in a decrease in bacterial biomass, which can be reduced by up to 30%. Soil is naturally prone to acidification when it rests on a granite, schist or sandy substrate. Acidification is also a natural process in temperate climates, which accelerates during the rainy season.
Over time, cultivated soils become more acidic as a result of ammonium nitrification caused by bacteria when synthetic nitrogen fertilisers (containing urea, ammonium nitrate, ammonium sulphate, etc.) or organic fertilisers (manure, compost) are added to the soil. It should be noted that potassium or sodium nitrate, dicalcium phosphate and calcium cyanamide have an alkalising effect when used alone. Acidification is also higher in winter.
A very humid and unventilated environment promotes the production of organic acids. Acidic soil is more prone to accumulating undecomposed organic matter, which facilitates the emergence of plant diseases.
Basic or acidic solution: what is the difference?
Any solution containing water and a dissolved substance is called an aqueous solution. All salts dissolved in water are in ionic form. Ionic aqueous solutions contain ions with a negative electric charge (anions) and ions with a positive electric charge (cations). A solution is neutral when it contains as many anions as cations. An excess of hydrogen cations H+ is responsible for the acidity of a solution. When hydroxide anions OH- are more numerous, the solution is basic (alkaline).
This diagram shows the impact of pH on nutrient availability and the emergence of toxicity in certain metals (particularly aluminium). Source: University of Nice-Sophia Antipolis

Optimal pH for absorption for some vegetables
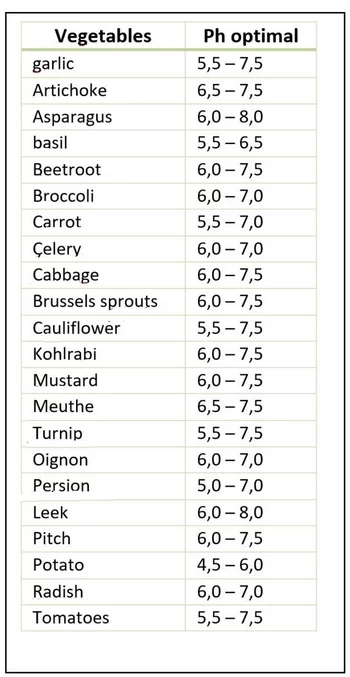
pH and absorption of nutrients present in the soil
The absorption of certain nutrients by plants depends on two factors related to soil pH
- The optimum soil pH for the absorption of each nutrient: Each nutrient has an optimum pH for availability to plants.
- The optimum pH for nutrient absorption for each plant: The optimum pH for absorption varies depending on the type of crop. For calcicolous crops, nutrient absorption is better when the pH is above 8. For acidophilic crops, a pH below 6.5 is preferable.
In agronomy, fertility potential is considered unaffected when the pH is between 6.5 (slightly acidic) and 7.5 (slightly alkaline). Most nutrients are optimally absorbed by plants within this pH range, which is also compatible with root growth. Microflora activity is more intense in a neutral environment. Near neutrality (pH 7), the microbial conversion of ammonia to nitrate is rapid. A neutral pH also promotes the degradation of plant protection products.
A pH below 5 causes excessive aluminium absorption, which is toxic to plants; a risk estimated at only 1% for French soils. (1)
The assimilation of nitrogen, potassium and sulphur by plants appears to be less affected by soil pH. However, the assimilation of phosphorus is more affected by soil pH. Most other nutrients (especially trace elements) tend to be less available when the soil pH is above 7.5, whereas they are perfectly available when the pH is slightly acidic, between 6.5 and 6.8. Molybdenum (Mo) is an exception, being less available at acidic pH levels and more available at moderately alkaline pH levels, which is very suitable for cucumber cultivation.
Soil pH plays an important role in ammonia volatilisation losses. A nitrogen fertiliser such as urea generally suffers greater losses at a pH of 8. However, there are other factors such as soil moisture, temperature, texture and cation exchange capacity that can affect ammonia volatilisation. The important point to remember is that under conditions of low soil moisture or poor fertiliser incorporation, volatilisation losses can be considerable even if the pH drops to as low as 5.5.
Soil pH is an important factor in nitrogen fixation in legumes. The survival and activity of Rhizobium bacteria (responsible for nitrogen fixation in association with legumes) decrease as soil acidity increases. This should be taken into account when attempting to grow legumes (beans, peas, etc.) on soils with a pH below 6.
In vegetable farming, a pH of 8 does not prevent acceptable harvests, including for potatoes, which in principle prefer slightly acidic soils. In the Durance Valley, there are many farmers who produce different varieties of potatoes even though their soil is rich in limestone with a pH of around 8. In general, these farmers have to deal with much more serious problems such as pest pressure (particularly Colorado beetles and wireworm larvae).
Taking into account all of the above factors, a neutral value is a good average for all crops. This is the value that amateur gardeners should aim for by adding appropriate soil amendments.
Beware of the alkalising effect of wood ash
Wood ash, which contains high levels of potash and calcium, has an alkalising effect. It acts very quickly, which must be taken into account when spreading it on crops (or adding it to compost). In regions where the soil is rich in limestone, some amateur gardeners spread wood ash in their gardens in the hope of improving soil fertility, even though their soil is already alkaline. Of course, this addition, which increases the soil’s pH, can have serious consequences for soil biodiversity and cause deficiencies in other elements. The amount of wood ash added should not exceed 15 kg per 100 m².
Tip :
Wood ash contains potash, which is highly soluble. It is therefore not advisable to spread it in winter. As soon as the first rains fall, the potash will be washed away into the groundwater. It is preferable to add wood ash by scratching it into the top few centimetres of soil just before planting crops.
pH measurement within the reach of the amateur gardener
The vinegar test is sometimes mentioned on websites (2) as a way of determining whether soil is acidic or alkaline. In reality, it mainly highlights the presence of limestone in the soil and does not provide any details on the pH value or the free limestone, also known as active limestone, present in the soil solution (not to be confused with total limestone, part of which is in solid form). It is not necessary to contact a laboratory every time you want to know the pH of the soil. There are electronic tools and chemical analyses available to amateur gardeners that can be used to measure the acidity or alkalinity of the soil.

Electronic pH testers suitable for agricultural activities
There are many models of pH testers available at all price points (they are also used in aquariums, swimming pools, etc.). Most of these testers must be calibrated using solutions. Some testers can be calibrated with a single solution (pH neutral), which is more than sufficient for agricultural use. The purchase price for a basic tester is around €40 (2018) including batteries and calibration solutions. For a slightly higher price, it is possible to purchase a model with replaceable probes, which are delivered calibrated. You can also order replacement batteries and bags of calibration solutions, which should normally be replaced every week. Do not purchase a tester from a shop that does not supply calibration solutions. A translation of the English manual for the AD100 and 101 testers is available here.
Laboratory pH paper suitable for agricultural activities
There are pH paper strips impregnated with a reagent that changes colour depending on the pH value available here
Tip
Regardless of the equipment chosen, great care must be taken when preparing the solution, as the analysis may be skewed if the following procedure is not followed:
- Do not use tap water, which is often not neutral. If it contains a little limescale, its pH will be alkaline. Distilled water must be used. Check the pH of the distilled water before starting your measurements. In principle, distilled water has a pH of 7, but it easily absorbs CO² and oxygen from the atmosphere, resulting in acidification. This acidification may already be present in unopened bottled water purchased in shops due to the industrial manufacturing process. This acidification is reversible; simply boil the water to allow the carbon dioxide to escape. Then return the water to a container and close the lid to prevent it from absorbing CO² again.
- Take soil samples from around ten different places in the garden. After mixing the different samples, place 100 to 150 g of soil in a small 300 ml jar, then add distilled water to the top of the jar. Shake to mix the distilled water thoroughly with the garden soil.
Correcting the pH of growing media
- In general, high pH levels are found in soils rich in limestone, while low pH levels indicate the presence of acidic organic substances in soils that are often low in limestone. Apart from mineral amendments designed to correct the structure of soil that is too calcareous or too clayey (such as clay or lime), which influence pH, the latter can be modified by various mineral or organic fertilisers or chemical substances.
- A high pH can be reduced by adding acidic organic inputs such as heather soil, seabird guano, blond peat, oak leaf compost or crushed pine bark, after checking that their pH is acidic.
- Peat and peat composts have a dual advantage. They are also known for their water retention capacity and high exchange capacity. The application rate for blond peat is approximately 5 to 10 litres per square metre. Peat-based soil improvers are prohibited in organic farming on the grounds that their exploitation is not sustainable and that peat bogs must be protected. Peat can be replaced by sphagnum fibres grown in former peat bogs, or even by coconut fibres or vermiculite for their absorbent properties
- To acidify the soil, you can also add a little sulphur. Sulphur oxidises to form sulphuric acid through the action of bacteria living in the soil. It takes about 0.4 kg of sulphur per 10 m² to lower the pH by one point. You can also spread iron sulphate, which works faster than sulphur. The amount of iron sulphate needed to reduce the pH by one unit also depends on the soil texture; it is approximately 0.5 kg per 10 m².
- Among kitchen waste, coffee grounds are known to acidify soil. A nitrogen-based mineral fertiliser, ammonium sulphate, also has the property of acidifying soil. Calcium cyanamide has the opposite effect.
- Caution: reducing the pH too much could seriously disrupt the microbial flora. Bacteria are more sensitive to acidic environments than fungi. Importing too much acid input leads to a loss of fertility.
- It is preferable to apply the treatment in several stages and then monitor the pH levels over several months. Sulphur treatments are rarely used in open fields due to their high cost. This treatment is more commonly used in horticultural production and private gardens.
- The addition of acidic substances does not produce acidification proportional to the volume of these substances. Below a certain volume, which varies depending on the nature of the soil, no significant change is observed. This is due to the buffering capacity of the free limestone present in the soil, which determines its ability to resist pH variations. The ability of a soil to maintain a constant pH is related to the volume of calcium and magnesium carbonate and bicarbonate ions present in the soil solution.
- Calcium carbonate is the main component of non-free limestone, often found in the crystallised forms of aragonite and calcite. Magnesium is most often present in the form of magnesite. In the presence of carbon dioxide from the atmosphere and biomass, calcium and magnesium carbonates, which are poorly soluble in water, are converted into bicarbonates that are highly soluble in water, defining its carbonate hardness.
- The higher the carbonate hardness, the more alkaline the water and the higher the pH. It is then more difficult to acidify the soil due to the buffering effect of the carbonate hardness. The greater the buffering capacity, the less the pH of the solution varies when an acidic substance is added. So don’t panic if adding specifically acidic organic matter does not immediately lower the pH of soil that is rich in free lime.
