Introduction to integrated methods in the vegetable garden
chapter crop sol
Other interesting data that may be included in a
laboratory analysis
Click on ♦ to go to a page in the chapter
Analysis of the physico-chemical properties of cultivated soils ♦
. Texture and structure of cultivated soils ♦
. Clay-humus complexes and cation exchange capacity ♦
⇒ interesting data that may be included in a laboratory analysis
Influence of pH on the fertility potential of cultivated soils ♦
Humus; formation and evolution ♦
Soil fertility: is the apocalypse coming tomorrow? ♦
The microbial world and soil fertility ♦
Rhizosphere, mychorizae and suppressive soils ♦
Correction of soils that are very clayey, too calcareous or too sandy ♦
Stimation of humus losses in cultivated soil ♦
Compost production for a vegetable garden ♦
Composting with thermophilic phase ♦
Weed management in the vegetable garden ♦
To plow or not to plow? ♦
The rotary tiller, the spade fork, and the broadfork ♦
Please share this website with your friends and family and anyone else who might be interested in integrated farming methods.
The Useful Water Reserve (UWR) of a cultivated soil
After intensive watering, water fills all the small cavities between the soil particles. After drying out, which allows dry work to be carried out, water remains in the microspores smaller than 30 microns, while the macrospores are filled with air. The retention point is the amount of water that a soil can store when it is dry. If there is no further watering, the soil continues to lose water.
Beyond a certain drying limit, plants are no longer able to extract water and begin to wilt. The usable reserve (or readily available reserve) is the amount between the water retention point and the point at which permanent wilting occurs. Water retention capacity varies depending on soil structure. The finer the soil texture, the higher the usable reserve (the more water the soil can hold). It is 80L/m3 for sandy soil and 300L/m3 for clay soil. A low retention capacity requires more frequent applications of soluble fertilisers to prevent them from being washed away. Knowing the available reserve also makes it possible to predict when watering is necessary and the periods during which it should be carried out.
Organic carbon content of a cultivation soil
The measurement of soil organic carbon is used to assess the total organic matter content of the soil. There are several methods of analysis, using combustion or humic pathways, to measure carbon content. The organic matter/carbon ratio is estimated to be roughly constant. Carbon represents only a fraction of the organic matter. In laboratory analyses, a multiplier factor is applied to take into account elements present in organic matter other than carbon, notably oxygen and nitrogen.
The organic matter content is obtained by multiplying the organic carbon content by a correction factor generally between 1.7 and 2, depending on the laboratory. The factor 1.7 is the most commonly used. The organic carbon content (Corg or C in some laboratory analyses) is expressed as a percentage of the soil analysed. Other laboratories apply different correction factors to convert the amount of organic carbon to the organic matter content depending on the nature of the soil. For example, for MOR-type humus soil, the correction factor can be between 1.77 and 1.93.
Measuring phosphorus reserves in cultivated soil
Different dosage methods are used depending on the pH of the soil. The Joret-Heber method consists of extracting assimilable phosphorus with ammonium oxalate in a neutral environment. The Joret-Heber method is considered satisfactory even though it gives a lower result (about 1/3) than the Dyer method, which is the most aggressive. With the Joret-Heber method, the optimum range is between 151 and 280, while with the Dyer method, the optimum range is estimated to be between 280 and 400. The Olsen method uses a reagent (sodium bicarbonate) that gives lower values than the two methods mentioned above. The report produced by the laboratory specifies the method used and when the values are exceeded, requiring correction.
Fertilisation; measurement of certain trace elements

Trace elements are measured in mg per kg of dry matter using the DTPA extraction method (AFNOR NF X 31-121 standard). If measurements show a trace element deficiency, this can be easily corrected using specific foliar fertilisers that act directly on the plants (a). Books and websites provide descriptions of trace element deficiencies based on simple visual observations, which is not the best method due to the risk of confusion between real and induced deficiencies. This is even more true when there are several deficiencies. Only laboratory analyses can accurately identify the existence and origin of one or more deficiencies.
a) Foliar fertiliser application is not very effective in dry weather.
To learn more about trace elements and fertilisation, click here.
Measuring some key elements of a growing medium
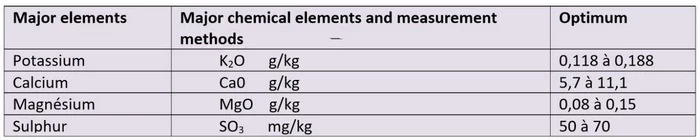
The various major elements are measured in g per kg of soil or in mg per kg of soil.
Fertilisation and specific potassium, magnesium and calcium ratios
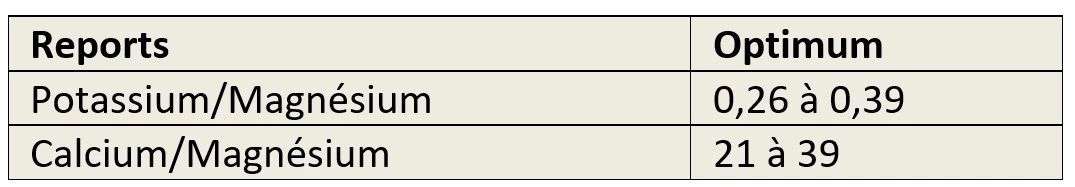
The measures must specify at least two ratios:
- Between potassium and magnesium,
- Between calcium and magnesium
in order to ascertain whether there is any risk of induced deficiency.
Cultivation soil, measurement of total and free lime
Measuring total limestone involves determining the amount of limestone present in the soil, including the fraction that is least soluble in water. Part of the limestone is easily attacked by carbonic acid present in rainwater or by organic acids in the soil to form soluble calcium bicarbonate, producing calcium ions (Ca²⁺). This fraction of limestone is called active or free limestone, which also includes other molecules such as calcium nitrate or monocalcium phosphates.
When Ca²⁺ cations are too abundant, they tend to saturate the absorbent complexes. Laboratory analyses can determine the amount of lime present in the soil solution and the amount of calcium ions bound by the absorbent complexes. However, active limestone is far from harmful as long as it remains incorporated in the soil water, as it maintains the coagulation of the clay. It is only when it comes into contact with the roots to form a suffocating sleeve that reduces iron absorption that limestone becomes toxic.
In arboriculture, knowledge of active limestone is essential for assessing the fruit-bearing capacity of soil. Tolerance standards are 15% for cherry trees, 10% for apple trees, 6 to 8% for pear trees and less for peach trees, which are the most sensitive fruit species (1). For vegetables, most of which are calcicolous, a soil rich in limestone is not a major concern, provided it has sufficient humus. However, adding organic matter to a very calcareous soil can have an adverse effect if the organic matter contains humic acids, which dissolve the limestone and increase its solubility. To avoid this problem, soil that is too calcareous must be amended with clay before organic matter is added.
Another form of limestone, gypsum (CaSO₄), promotes plant growth when its content in the soil is between 2 and 25%. Above this level, gypsum can cause significant reductions in crop yields. In soils with a high active limestone content, any additions that could increase this level should be avoided if it is not suitable for the crops being grown.
Above 5% total limestone, resulting in systematically alkaline soil, natural calcium reserves and their gradual release through dissolution make rapid limestone supplementation such as liming unnecessary. Determining the active limestone content allows the Chlorosis Power Index (CPI) to be defined, which is an estimate of the risk of iron deficiency.
Soil that is low in free iron and contains active limestone can result in poor iron assimilation (iron chlorosis). Certain plants such as strawberries, vines and some fruit trees such as apple trees are sensitive to this type of deficiency. When active limestone is less than 4%, the risk of iron chlorosis is very low. An active limestone content of more than 6% induces a significant risk of iron chlorosis, which becomes very high when the content exceeds 10%.
Other measures
Depending on the type of accreditation granted, some laboratories may produce other analyses, particularly on trace elements (cadmium, chromium, copper, nickel, lead, zinc, mercury) or use more precise methods for sampling and storing soil samples in order to determine the mineral nitrogen content of fresh soil. These analyses should be undertaken when it is suspected that recently acquired agricultural soil contains pollutants, or when there is a desire to simply determine the content of certain trace elements.
1) Le thème de l’eau dans la vallée du Rhône – essai sur la genèse d’un espace hydraulique – J Bhetemont – 1972
